
^Powered by the Arbitrary Illumination Microscopy with Encoded Depth (AIMED) technique, axial encoding enables simultaneous multiplexing of multiple depth layers, allowing volumetric information to be reconstructed from substantially fewer scans through the joint use of structured illumination and compressive sensing.
The Speed Challenge in Multiphoton Microscopy
Multiphoton microscopy (MPM) is a cornerstone technique for deep-tissue three-dimensional imaging in life sciences, playing an indispensable role in in-vivo studies of neuronal structures, vascular networks, and functional dynamics. However, acquiring a full 3D volume with conventional MPM typically relies on sequential axial scanning, where only a single focal plane is imaged at a time. This approach suffers from low imaging efficiency and high cumulative light exposure, significantly limiting its applicability to fast biological dynamics and long-term observations.
Over the past decade, various strategies have been explored to overcome this bottleneck, including non-mechanical axial focusing, temporal delay encoding, and beam multiplexing techniques. Despite notable progress, these methods face upper limit of the achievable maximum scanning rate restricted by full sampling.
To address these challenges, a fundamentally new imaging strategy in a sub-sampling way was developed by researchers from the OMEGA lab by Professor Kenneth K. Y. WONG from the Department of Electrical and Electronic Engineering, The University of Hong Kong. This framework is termed AIMED. By combining innovations in axial optical encoding and computational image reconstruction, AIMED achieves a substantial enhancement in 3D imaging speed and photonic safety with minimal system overhead, demonstrating comprehensive advantages in efficiency, image quality, and system compatibility.
Axially Encoded Multiplane Imaging Strategy
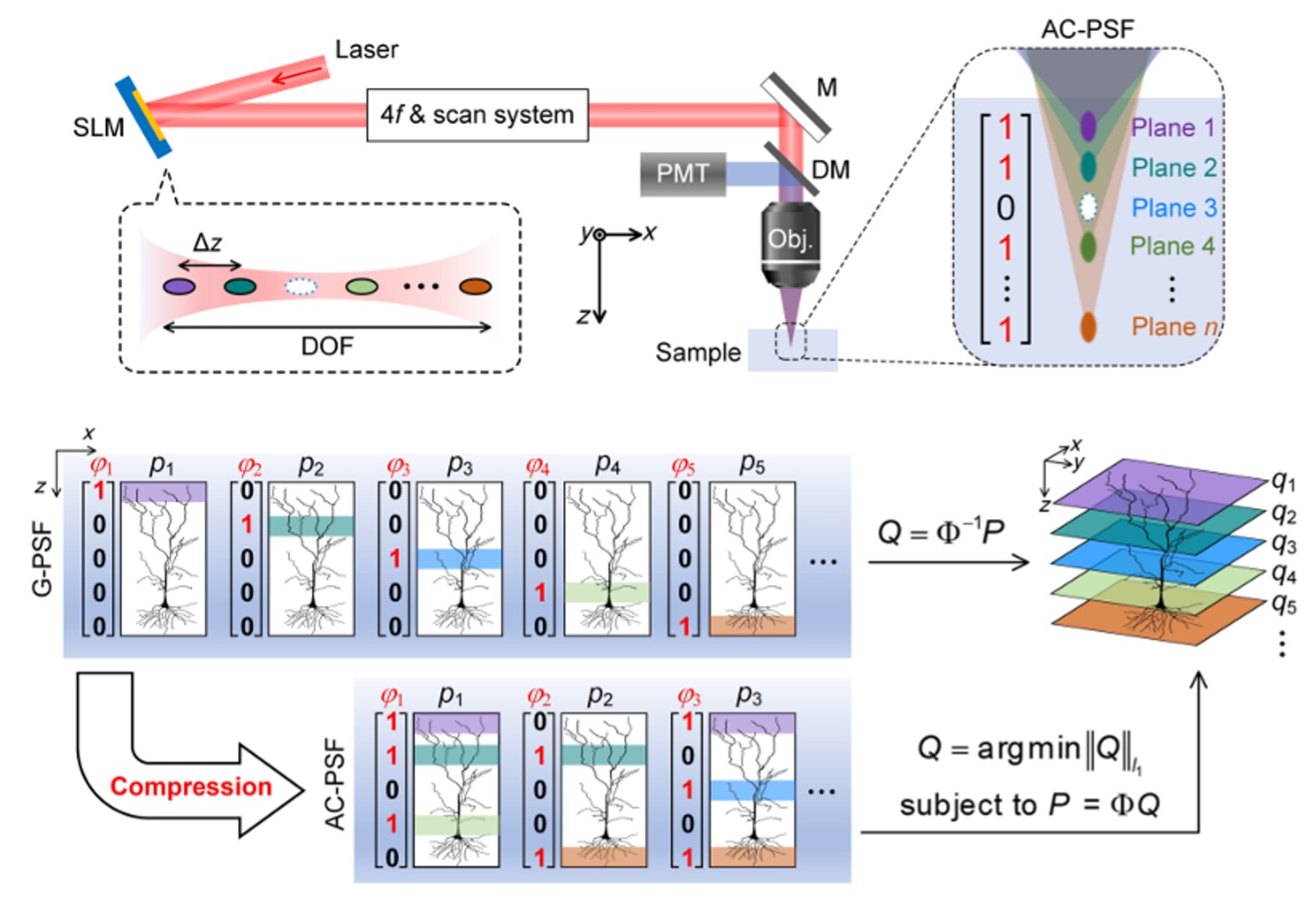
The core concept of AIMED departs from the conventional paradigm of plane-by-plane scanning. Instead, it employs axially structured illumination to simultaneously excite multiple depth layers within a single exposure, followed by computational reconstruction based on compressive sensing principles.
On the optical side, the research team uses a spatial light modulator (SLM) to load designed phase masks that split an incident laser beam into multiple controllable focal spots along the propagation direction. These focal spots can be flexibly combined following Hadamard encoding schemes, forming programmable axial on/off (0/1) illumination patterns. Moreover, the relative intensity of each focal spot can be independently adjusted to compensate for depth-dependent attenuation or signal imbalance.
When interacting with the sample, the nonlinear nature of two-photon or three-photon excitation naturally suppresses inter-plane crosstalk, enhancing the independence of the encoded layers. On the imaging side, instead of sequential axial scanning, only a limited number of encoded illuminations are required. Depth-resolved fluorescence signals are then recovered using sparse optimisation algorithms, enabling full 3D reconstruction from compressed measurements.
Notably, AIMED does not require modifications to the laser source, scanning optics, or detection modules. The implementation only involves inserting an SLM and a compact 4f optical system into the excitation path, offering excellent system universality and deploy ability across existing multiphoton platforms.
Validation in Mouse Brain Multiplane Imaging
The multiplane focusing and encoding performance of AIMED was first validated through simulations and experimental characterisation. Axial-coded point spread function measurements under different encoding schemes demonstrate precise axial control and good intensity uniformity across multiple planes. In a five-plane configuration, the lateral resolution remains around 600 nm, while the axial resolution ranges from 2 to 4 mm, indicating that high-quality optical focusing is preserved even under simultaneous multilayer excitation.
AIMED was further evaluated in imaging experiments on mouse brain neuronal samples. Compared with conventional plane-by-plane scanning, AIMED successfully resolved fine neuronal substructures, including dendrites and axons, under a compression ratio of approximately 60%, while using only one-half to one-third of the per-plane optical power. In some encoding configurations, the reconstructed images even exhibit enhanced contrast.
For particularly delicate structures such as dendritic spines, AIMED consistently delivers reconstruction fidelity comparable to or better than traditional high-power sequential scanning.
High-Fidelity Reconstruction under Strong Compression
The robustness of AIMED under different compression ratios was systematically examined. Across compression ratios ranging from 62.5% to 87.5%, the reconstructed 3D images maintain a structural similarity index of approximately 0.95 and a peak signal-to-noise ratio of 41-42 dB, showing negligible degradation compared with fully sampled volumetric scans.
Further numerical studies indicate that, in large-scale volumetric tasks involving up to 47 axial planes, AIMED can achieve an approximately eightfold increase in acquisition speed, highlighting its strong scalability and potential for high-throughput volumetric imaging.
Technical Advantages and Future Perspectives
The paradigm embodied by AIMED, axial optical encoding combined with sparse reconstruction, provides a lightweight, flexible, and efficient solution for 3D multiphoton imaging. Unlike hardware-intensive acceleration strategies, AIMED does not rely on expensive components or major system reconfiguration. Instead, it leverages programmable light-field engineering together with a mature compressive sensing framework to improve imaging speed while preserving image fidelity and system stability.
This approach is particularly well suited for sparse biological structures such as neuronal networks and is inherently favorable for phototoxicity-sensitive samples. Looking ahead, the principles and framework of AIMED are readily transferable to other three-dimensional optical imaging modalities, including confocal microscopy, Raman imaging, and photoacoustic imaging.
By enabling faster, deeper, and longer-term volumetric imaging, AIMED also lays a foundation for future integration with data-driven and deep-learning-based intelligent imaging strategies.
Read more in the article:
- Xin Dong, Hongsen He, Minghui Shi, Cora S. W. Lai, Kevin K. Tsia, and Kenneth K. Y. Wong. “Multiplane compressive imaging with axial-coded multiphoton microscopy.” Advanced Photonics 7, no. 4 (2025): 046010-046010.
https://doi.org/10.1117/1.AP.7.4.046010
Other related publications:
- Hongsen He; Minghui Shi; Beth Wing Lam So; Qiyu Zheng; Yuanjia Wang; Cora S. W. Lai, Kevin K. Tsia, Xin Dong, Kenneth K. Y. Wong. “Axially Parallel Excitation with Depth Encoding in Multiphoton Microscopy Enables High-Speed, High-Contrast 3D Imaging of Brain Neurons,” 2025 IEEE Photonics Conference (IPC), Singapore, 2025, pp. 1-2, doi: 10.1109/IPC65510.2025.11281887. (Post-deadline paper)
- Hongsen He, Huajun Tang, Meng Zhou, Hei Ming Lai, Tian Qiao, Yu-Xuan Ren, Cora S. W. Lai, Ho Ko, Xiaoming Wei, Zhongmin Yang, Kevin K. Tsia, Kenneth K. Y. Wong. “Deep-tissue two-photon microscopy with a frequency-doubled all-fiber mode-locked laser at 937 nm.” Advanced Photonics Nexus 1, no. 2 (2022): 026001-026001.
- Hongsen He, Xin Dong, Yu-Xuan Ren, Cora S. W. Lai, Kevin K. Tsia, and Kenneth K. Y. Wong. “Tomographic-encoded multiphoton microscopy.” ACS Photonics 9, no. 10 (2022): 3291-3301.
Related news:
